13. Enthalpy and entropy changes of a reversible reaction are 40.63 kJ mol–1 and 108.8 J K–1 mol–1 respectively. Predict the feasibility of the reaction at 27^° C.

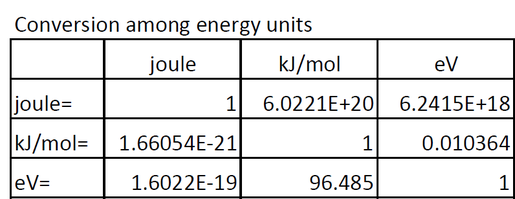
enthalpy - Why is it sometimes kJ only, and in other times kJ/mol? What's the difference? - Chemistry Stack Exchange

Calculated Free Energies (G, kJ/mol) and Vibrational Frequencies (ν̃ ,... | Download Scientific Diagram
The activation energy of a reaction is 75.2 kJ mol^-1 in the absence of a catalyst and it lowers to 50.14 kJmol^-1 with a catalyst. - Sarthaks eConnect | Largest Online Education Community

∆H 0 m (kJ mol −1 ) and ∆S 0 m (kJ mol −1 K −1 ) for the SDS-caffeic... | Download Scientific Diagram

Enthalpy of formation of H,O, is: (1) +X, kJ mol-1 127-X, kJ mol (3) +X, kJ molt (4) -X, kJ mol Given that bond energies of H-Hand CHCl are 430 kJ moll

SOLVED: Given the data: N2(g), ΔH° = 0.00 kJ mol-1, ΔS° = +191.5 J mol-1 K-1 Hz(g), ΔH° = 0.00 kJ mol-1, ΔS° = +130.6 J mol-1 K-1 NH3(g), ΔH° = -46.0

enthalpy - Why is it sometimes kJ only, and in other times kJ/mol? What's the difference? - Chemistry Stack Exchange

43. Energy of the electron in Hydrogen atom is gives by (a) En2 kJ mol- (b)En kJ mol- (c) En = (d) En = 131.38 131.33 1313.3 _ kJ mol-1 313.13 kJ mol-
According to the following figure, the magnitude of the enthalpy change of the reaction A + B → M + N in kJ mol^–1 - Sarthaks eConnect | Largest Online Education Community








![Solved Given that ??1f[H(g)]-218.0 kJ . mol-1 ??;[dg)]-716.7 | Chegg.com Solved Given that ??1f[H(g)]-218.0 kJ . mol-1 ??;[dg)]-716.7 | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2Faee%2Faeef339b-3db2-4a6c-bb5a-a95b061f7a49%2FphpqsRWV0.png)


![Solved Given that Delta H degree r[H(g)] = 218.0 kJ. mol-1 | Chegg.com Solved Given that Delta H degree r[H(g)] = 218.0 kJ. mol-1 | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media/f73/f73ff219-6265-444d-89c1-620f5d2f2822/php7wff8s.png)